Moreover, the content of this review appraises computational efforts, such as DFT quantum mechanical modelling, in developing mechanistic (i. e., both thermodynamic and kinetic) insights into the reaction of singlet oxygen with hydrocarbons. The review assembles critical scientific studies on the implications of singlet oxygen in initiating chemical reactions, identifying, and quantitating the consequential effects on combustion, fire safety, as well as on the low-temperature treatment of organic wastes and contaminants. This paper reviews the intrinsic properties of singlet oxygen, emphasising the physical aspects of its natural occurrences, production techniques, as well as chemical reactivity with organic fuels and contaminants. This book presents information about the high enzymatic reactivity of reactive oxygen species. Such reactivity of singlet oxygen has inspired a wide array of emerging applications in chemical, biochemical and combustion phenomena. 9. 
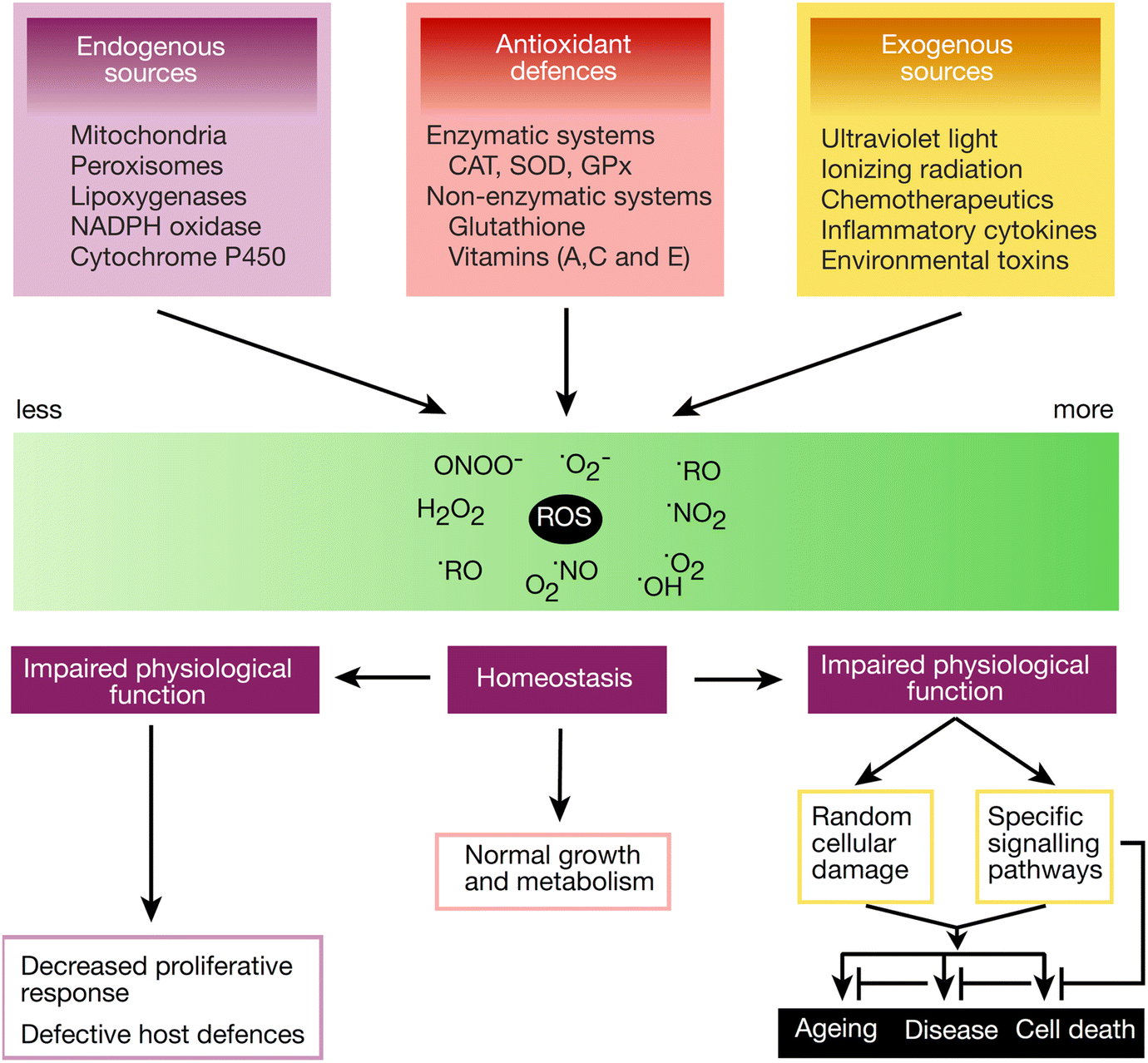
Singlet oxygen has a relatively high energy of about 94 kJ/mol compared to the ground state molecular O 2 and therefore initiates low-temperature oxidation of electron-rich hydrocarbons. 
Pure graphene has excellent electronic and mechanical properties and it is cheap to produce, thus making it a great potential candidate for green technology, which is represented by the ivy. The less reactive metals such as sodium potassium and lithium are stored in. By moving down the group reactivity is increased. In the gas phase the dramatic example of oxygen radical reactivity with hydrocarbon substrates is combustion, which is initiated by HO(or ROor MO) and. They rapidly react with oxygen so they should be stored out of contact with oxygen to prevent the oxidation process. I Departamento de Química, Universidade de Évora, Largo dos ColegiÉvora, Portugal. Cristina Sousa I Ana Maria Botelho do Rego II Teresa Sá e Melo II, e-mail:. Group I consist of alkali metals and these are very reactive. Singlet oxygen reactivity in water-rich solvent mixtures. In general, highly reactive oxygen-bearing molecules remain the backbone of diverse ground-breaking technologies, driving the waves of scientific development in environmental, biotechnology, materials, medical and defence sciences. A radical (often, but unnecessarily called a free radical) is an atom or group of atoms that have one or more unpaired. The Front Cover picture shows the oxygen reactivity at the interface between graphene and a copper foil. Reaction of Group I Elements with Oxygen. given it the fulminating cap that initiates the reactivity and accelerates it. Singlet oxygen represents a form of reactive oxygen species (ROS), produced by electronic excitation of molecular triplet oxygen. Molecular oxygen is very chemically reactive, and tends to form oxides by reaction with other elements and compounds quite easily. Molecular oxygen is not reactive, but add to it small amounts of active.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
